Clomipramine is a widely available tricyclic antidepressant, which can be repurposed to treat brain tumours or brain cancer and can improve the performance of Temozolomide with GBM and enhance survival times, according to Professor Geoffrey Pilkington.
By Professor Geoffrey Pilkington BSc PhD CBiol FIBiol FRCPath
Professor of Cellular & Molecular Neuro-oncology, School of Pharmacy & Biomedical Sciences, University of Portsmouth, and Professor of Experimental Neuro-oncology, Kings College, London.

Professor Pilkington has been involved in brain tumour research since 1971 and has published numerous scientific papers, particularly in the field of neurocarcinogenesis and brain tumour cell invasion into the normal brain. After 23 years at Kings College, London, Professor Pilkington recently left his post as Professor of Experimental Neuro-oncology to take up a new post at the School of Pharmacy & Biomedical Sciences of the University of Portsmouth.
He is currently involved in clinical trials (supported by the Samantha Dickson Research Trust) using the tricyclic drug, clomipramine, as a putative anti-tumour agent in malignant brain tumours. He has served on committees of the British Neuropathological Society, the European Association for Neuro-oncology, the British Neuro-oncology Group and has organized numerous brain tumour conferences, both national and international. He is currently a co-ordinator of the International Glioma Invasion Forum series and contributes lectures on the structure and function of the brain and on the biology of brain tumours to diverse course run by various UK universities. In the mid-1990s he catalysed the formation of UKBTS by bringing together a number of brain tumour charities and individuals with the common goal of facilitating improved research and treatment strategies. He is also a member of the Scientific & Medical Advisory Boards of the Samantha Dickson Research Trust, Charlies Challenge and Ali's Dream. In his "free" time he is a keen cricketer & rugby enthusiast.
Clomipramine: A Breakthrough For Brain Tumours?
In more than 30 years laboratory-based research into the biological nature of brain tumours, few developments have excited me more than the seemingly bizarre finding that an antidepressant drug may provide an effective way to combat this resistant group of cancers.
The major types of tumours, which begin from the substance of the brain, are known collectively as gliomas. These may arise as high-grade malignancies or may be low grade, benign tumours. Unfortunately the so-called benign tumours frequently develop into malignant tumours with the passage of time. The devastating news that patients with the most malignant form of glioma - known as glioblastoma multiforme - are faced with is that in the absence of treatment they are likely to live for only a few months. Even with surgery, radiotherapy and possibly cytotoxic chemotherapy the 5-year survival rate is very poor. The use of cytotoxic drugs in this group of tumours also remains controversial since the modest survival benefit claimed by some workers has to be tempered by the probable reduction in quality of life.
Sadly, despite all the advanced imaging and surgical technology and genetically based approaches to therapy there has been little to encourage either patients or clinicians in the fight against malignant glioma over the past 40 years. Now, following several years laboratory research, trials will finally begin for anaplastic astrocytoma and glioblastoma multiforme (the two most malignant forms of primary brain tumour in adults) using a drug which has been known to us throughout these past 4 decades - as an antidepressant!
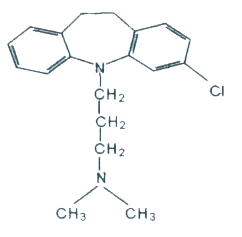
Fig 1 Chemical structure of clomipramine
I was first introduced to the concept of the antidepressant, clomipramine, as a potential anti-cancer agent through a small scrap of University College, London headed paper from Professor David Wilkie. David, who is a well-respected retired Professor, had simply written that he had read my papers on brain tumour cell invasion and thought he had something that might interest me. In short we met and David explained how, through his early studies with yeast cells, he had shown that the tricyclic drug imipramine, and its more active chlorine derivative, clomipramine (or chlorimipramine) influenced energy metabolism and that cancer cells might, at least in theory, be combated by this agent.
Go To: Alternative brain cancer treatments that worked!
If clomipramine was able to act selectively on cancer cells due to their compromised respiratory function, as appeared to be the case, then this drug was indeed to be applauded. Clomipramine is one of a group of tricyclic drugs, a name based on their chemical structure (fig 1). Its major use had been in treatment of obsessive/compulsive and clinically depressed patients. As far as the seemingly strange possibility of treating brain tumours with such an agent was concerned, it did have a number of very attractive features. Notably, after oral administration it is stored in high quantities in both the brain and lung and it has a long half life of approximately 24 hours which is increased to 40 hours once metabolized in the body to its byproduct, desmethyl clomipramine. The longer the half-life, the longer before the agent will break down. The very fact that the drug crosses the so-called "blood-brain barrier" (which is formed by specialized blood vessels within the brain) and therefore enters the normal brain is a great advantage in the treatment of brain tumours. Many of the cytotoxic drugs used get into the major mass of the brain tumour by virtue of the fact that the barrier is broken down within the tumour but at the tumours edge many cancer cells migrate or "invade" into the normal brain and are protected from these cell killing drugs by the "blood-brain barrier".
There has been little to encourage either patients or clinicians in the fight against malignant glioma over the past 40 years
We first started to work on the action of clomipramine on brain tumour biopsies in the form of an MSc student project over a 6-month period. Here, both the student, Emma Daley, and I were surprised and delighted to note that, while the drug killed cancer cells, in a dose dependent manner, normal brain cells were completely unaffected. We also looked at a number of other agents with a very similar chemical structure notably chlorpromazine (a tranquilizing drug used extensively in the treatment of psychosis) and clofazimine (an anti-leprosy drug) but these either failed to kill tumour cells or caused only reversible damage, killing merely a proportion of component cells of the tumour. On the basis of these early studies I submitted a grant application to the Samantha Dickson Research Trust for funding of a more in depth analysis of how the drug may work and which tumour types might be sensitive. We were lucky enough to receive 3 years support from the Trust and recruited the all important help and collaboration of Dr Tim Bates a senior mitochondrial biochemist from the Institute of Neurology in London. After just a few assays where the action of clomipramine was compared with standard inducers of cell death Tim was just as excited as we were at what he was seeing.
Unlike most cytotoxics, which act by damaging the DNA within the nucleus of the cells, clomipramine acts via cellular respiratory centres, called mitochondria, which are responsible for production of energy for the cells various activities. The mode of action is therefore, different in that it promotes natural cell death (called apoptosis or programmed cell death) due to compromised respiratory function in cancer cells while leaving normal cells unaffected. In essence, the drug enters the mitochondria affects complex III of the respiratory chain, reduces oxygen consumption, thereby increasing reactive oxygen species and liberating cytochrome c as a consequence. This, in turn activates a series of enzymes which result in programmed cell death. On the basis of these experimental studies last year Emma Daley was awarded the degree of Doctor of Philosophy by the University of London.
Interestingly, Professor John Gordon of the University of Birmingham has recently found clomipramine to have a similar action on lymphoma cells and has also noted that another group of antidepressants, the selective serotonin reuptake inhibitors (SSRIs), appear to elicit similar effects.
The side effects are generally mild and include tiredness and dryness of the mouth
Although the clinical trial has not yet officially begun, to date, some 250 "anecdotal" cases have been treated. Although the longest standing glioblastoma patients have only been receiving clomipramine for 3 years, there have been numerous reports of reduction on tumour mass on MRI scanning, increased survival times, marked clinical improvement and improved quality of life over and above those treated with surgery and radiotherapy alone or with adjuvant cytotoxic chemotherapy. Moreover, benefit to patients who had failed both radiotherapy and cytotoxic chemotherapy, has been apparent.
Go To: American Scientist finds parasites in all cases of brain cancer
The side effects are generally mild and include tiredness and dryness of the mouth (which may reduce with time). Since the drug lowers the threshold for seizures in patients whose tumours are likely to give rise to such events, the level of anti-convulsant treatment should be carefully monitored. It is also important for patients to inform their doctors if there are any additional possible side effects such as palpitations or difficulty in passing urine. Patients on drugs known as monoamine oxidase inhibitors will not be allowed to take clomipramine. Since the drug has been widely used drug over 35-40 years there is more than adequate information on toxicity and therefore a phase I trial is not necessary. Administration is orally as either capsules or tablets but, unfortunately, clomipramine syrup has been recently withdrawn due to low usage.
The question arises of when or whether to stop treatment if the tumour shows no clinical or MRI/CT scan based evidence of progression. Unfortunately, at this stage we do not have a real answer but, if patients do come off it, it is recommended that this is done slowly in gradual increments. Our suggestion is, however, to stay on the drug long term and only if neuro-imaging shows sustained and total response in terms of inactivity of tumour should cessation of drug use be considered.
We are currently exploring ways of enhancing the action of clomipramine and other pro-apoptotic drugs
The research work does not end here, however, and I would not claim that clomipramine is in itself a "gold bullet" glioma cure all. Nor would I claim that it is suitable for the treatment of all brain tumours. Indeed, we are currently exploring ways of enhancing the action of clomipramine and other pro-apoptotic drugs. To these ends we have recently shown that certain enzymes, such as cathepsin L, which are present in high amounts in gliomas and increase with grade of malignancy, protect tumour cells against apoptosis. Therefore, inhibitors of these enzymes may be of clinical use. We are now investigating such inhibitor systems in collaboration with Professor Tamara Lah of the Slovenian National Institute of Biology, Ljubljana. In addition, our studies have gained considerable support from Dr Robert Jones, who has, over the years, made an in depth study of the role of tricyclic drugs and dietary supplements in the treatment of cancer. Dr. Jones reports that the suspicion that certain polyunsaturates participate in the tumour cell killing process indicates that supplementation of clomipramine therapy with this omega 3 oils may be of value.
Go To: Complementary and alternative brain tumour treatments
Perhaps the most frustrating thing about using a drug such as clomipramine which is relatively inexpensive and now out of patent is that the major pharmaceutical companies show little interest in is novel use as an anti-cancer agent. If we could develop a new drug with a similar structure and activity and, more significantly, greater marketing potential we would perhaps be in a better position.
The clinical trial - supported by the Samantha Dickson Research Trust - will begin in June at Kings College and St Thomas' Hospitals for patients between 16 and 65 years of age with newly diagnosed and histologically verified anaplastic astrocytoma or glioblastoma multiforme. It is hoped that the trial will be extended to other centres in the UK and that if further funding can be provided with support from paediatric oncologists it may be possible to extend the trial to children suffering from brain stem glioma.